My primary research interests are in the understanding of the biology of insect pests and their interactions with other insects, plants, and the environment and development of integrated pest management strategies for existing and invasive agricultural insect pests.
Experiences and expertise are in conducting laboratory, growth chamber, greenhouse, field cage, and open field experiments and in using research tools such as camera-mounted microscope, ImageJ software, and leaf area meter, and statistical softwares such as JMP, SAS, and R.
My recent research experiences are in the assessment of the abundance, distribution, and movement of economic insect pests along with development of thresholds and sampling plans, and determination of their host preference, survivability, and growth response on host plants. Experiences and interests are also in the evaluation of biological and chemical control options.
Crops of interest include vegetable, ornamental, fruit, and field crops. The ultimate research goal is to improve the understanding of insect biology, develop IPM tactics, and reduce insects pests related crop loss.
Below are the research projects I conducted or participated in:
Experiences and expertise are in conducting laboratory, growth chamber, greenhouse, field cage, and open field experiments and in using research tools such as camera-mounted microscope, ImageJ software, and leaf area meter, and statistical softwares such as JMP, SAS, and R.
My recent research experiences are in the assessment of the abundance, distribution, and movement of economic insect pests along with development of thresholds and sampling plans, and determination of their host preference, survivability, and growth response on host plants. Experiences and interests are also in the evaluation of biological and chemical control options.
Crops of interest include vegetable, ornamental, fruit, and field crops. The ultimate research goal is to improve the understanding of insect biology, develop IPM tactics, and reduce insects pests related crop loss.
Below are the research projects I conducted or participated in:
Chilli thrips
Chilli thripsPhD Research (2016-2020):
Ecology, behavior, and management of chilli thrips, Scirtothrips dorsalis Hood (Thysanoptera: Thripidae) in Florida strawberry and blueberry. Advisor: Dr. Justin Renkema, Research Scientist (Agriculture and Agri-Food Canada) Co-advisors: Dr. Oscar Liburd, Professor and Dr. Sriyanka Lahiri, Assistant Professor (University of Florida) |
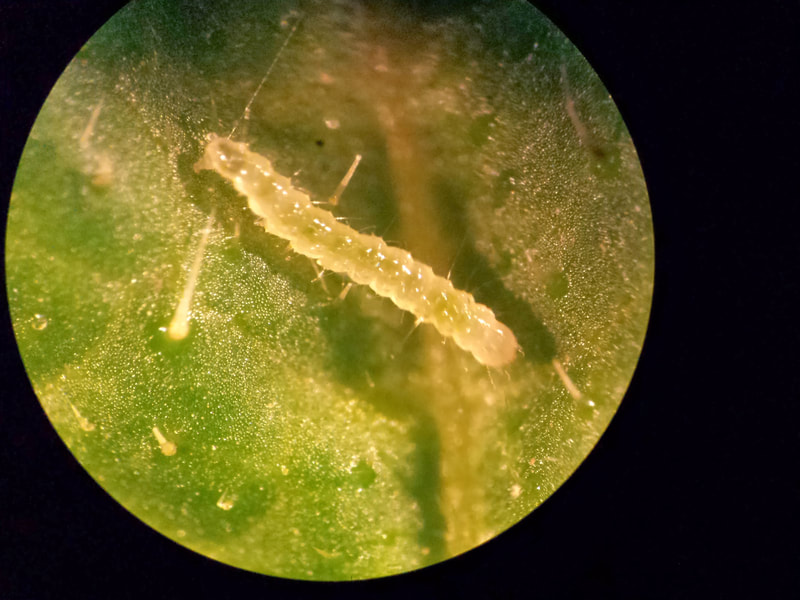
Chilli thrips is an invasive and foliar pest of vegetable, ornamental, and fruit crops in southeastern United States. This pest cause damage by feeding or plant damaging viruses. Although, chilli thrips is an efficient vector of tospoviruses, there are no such reports in crops grown in United States. In central Florida, chilli thrips is an important foliar pest of strawberry and blueberry. In strawberry fields, chilli thrips immigrate from nearby crop or vegetation after newly transplanted strawberry plants bear new leaves. Adult and larval chilli thrips cause injury to new leaf growths by removing cellular contents using their piercing-sucking mouthparts. Heavy infestations of chilli thrips cause stunted plant growth and reduced yield. Due to limited information on threshold and management of this pest in Florida berry crops, control decisions that are made might be inaccurate or unnecessary. Greenhouse, field plot studies, and field surveys were conducted during 2016-2019 to study the ecology, behavior, and management of chilli thrips in Florida strawberry and blueberry.
Research projects:
- Conducted greenhouse and field plot studies to determine the short-range movement of chilli thrips and rate of spread of feeding injury among strawberry plants.
- Conducted field plot studies to determine if delayed insecticide application is useful in managing chilli thrips in strawberry.
- Surveyed strawberry and blueberry fields to assess the spatial and temporal distribution of chilli thrips.
- Conducted field cage studies to determine the critical density (proxy of action threshold) of chilli thrips in strawberry.
- Developed a sequential sampling plan of chilli thrips in strawberry and a fixed-precision sampling plan in blueberry.
- Tested the efficacy of a reduced-risk butenolide insecticide, flupyradifurone to control chilli thrips in strawberry.
- Conducted field trials to test the efficacy of various insecticides against chilli thrips in strawberry and blueberry.
Results showed that chilli thrips move slowly between strawberry plants. Due to the slow movement, chilli thrips showed temporally stable aggregated distribution within strawberry fields. Moreover, actively growing strawberry plants compensated for the minor injury with no reduction in plant vigor and yield when when treated with insecticide after two weeks of initial infestation of five to 20 thrips per plant. Since, chilli thrips remain in initially infested plant for two weeks and initial five to 20 thrips per plant do not reduce yield when treated after two weeks, management actions can occur in two weeks interval. The critical density of chilli thrips was established as two thrips per trifoliate which is least number of thrips that can reduce yield if left untreated for three weeks. Management decisions of chilli thrips in strawberry can now be made using a newly developed sequential sampling plan. To make a decision, collect at least four sampling units from random locations in field. One sampling unit is one strawberry trifoliate from each ten adjacent plants. Compare thrips counts with lower and upper limit of the sequential sampling unit. If thrips count is greater than upper limit, treat the field; if between upper and lower limits, continue sampling, and if total thrips count is below lower limit, do not treat and resample in two weeks. Spinetoram is most effective in controlling chilli thrips in strawberry and blueberry. Other fairly effective insecticide against chilli thrips are cyantraniliprole and flupyradifurone in strawberry and tolfenpyrad in blueberry, and can be used in insecticide rotation program.
In blueberry, a fixed-precision sampling plan was developed which can estimate the field mean population of chilli thrips with fewer samples as oppose to whole field scouting.
In blueberry, a fixed-precision sampling plan was developed which can estimate the field mean population of chilli thrips with fewer samples as oppose to whole field scouting.
Pictures of chilli thrips, its feeding damage, experimental layout, and materials and methods from research projects conducted to study the biology of chilli thrips in strawberry and blueberry
(Hover the mouse over image for caption)
(Hover the mouse over image for caption)
Flower thrips
In Florida, western flower thrips, Frankliniella occidentalis (Pergande), and Florida flower thrips, F. bispinosa (Morgan), occur in southern highbush, Vaccinium corymbosum L. × V. darrowi Camp (Ericaceae), and rabbiteye, V. ashei Reade, blueberry fields during flowering stage. Florida flower thrips is native to Florida and is more abundant than western flower thrips in blueberry. Both flower thrips can cause damage to blueberry fruits by ovipositing and feeding on the ovaries. One field plot study was conducted at Plant Science Research and Education Unit, University of Florida, Citra, FL during March to April, 2016 in rabbiteye blueberry planting. Experiment evaluated the effectiveness of two important natural enemies of flower thrips in blueberry: a predatory phytoseiid mite, Amblyseius swirskii Athias-Henriot (Acari: Phytoseiidae), and minute pirate bug, Orius insidiosus Say (Hemiptera: Anthocoridae) in rabbiteye planting. Natural enemies were released after the onset of flowering and occurrence of thrips population in blueberries. The release rates for both natural enemies (treatments) were heavy curative. Commercially available products used in the study with their release rates were: Thripor-I (O. insidiosus) @ 10 insects per m^2, and Swirski-Mite (A. swirskii) @ 100 insects per m^2.
Melonworm
Melonworm is a serious pest of cucurbitaceae throughout the southeastern United States. The adult moth is active throughout the winter months in southern Florida and disperses throughout the southern and gulf coast states every summer. Third through fifth instar melonworm larvae feed voraciously on the whole plant including fruit, leaves, and stalks; often removing leaf material while leaving veins and veinlets intact. Field plot and laboratory studies were conducted at research facility of Tropical Research and Education Center, University of Florida, Homestead, FL during 2013-2015 to better understand the biology and population dynamics of this pest in cucurbits.
Research projects:
- Field plot studies to assess the seasonal abundance and spatial distribution of melonworm in yellow squash.
- Field plot studies to determine the host preference and within-field distribution of melonworm in yellow squash, zucchini, cucumber, and watermelon.
- Choice and no-choice tests to determine the host selection of melonworm larvae among four cucurbit crops.
- Laboratory studies to determine the survivability and development time of larval and pupal stages of melonworm when fed with leaves of yellow squash, zucchini, cucumber, and watermelon.
- Measurement of head-capsule width and whole body length of melonworm larvae to determine the growth response of melonworm when fed with leaves of yellow squash, zucchini, cucumber, and watermelon.
Spotted Wing Drosophila
Drosophila suzukii Matsumura (Diptera: Drosophilidae), also commonly known as Spotted Wing Drosophila (SWD), is a pest of thin-skinned fruits worldwide. Since its invasion in 2008, SWD has become a significant pest of berry and cherry crops in the US, with economic losses amounting to millions of dollars.
Insecticide efficacy trials
Chilli thrips
Strawberry (2019)
Advisor: Sriyanka Lahiri, Assistant Professor (University of Florida)
Several insecticides were compared for efficacy in the field for management of chilli thrips at the research facilities of the Gulf Coast Research and Education Center, University of Florida, Wimauma, FL in 2019.
Products tested: Apta (tolfenpyrad, IRAC code #21A) at 27 oz/acre, Exirel (cyantraniliprole, IRAC code #28) at 16 oz/acre (low) and 20.5 oz/acre (high), Minecto Pro (abamectin, IRAC code #6 and cyantraniliprole, IRAC code #28) at 10 oz/acre, and Radiant (spinetoram, IRAC code #5) at 10 oz/acre. All experimental products were tank mixed with a surfactant, Induce, at the rate of 0.25% (v/v).
Results are published in AMT. Link to pdf
Blueberry (2019)
Advisor: Oscar Liburd, Professor (University of Florida)
Several insecticides were compared for the efficacy in two blueberry fields, located in Plant City, FL and Clermont, FL with southern highbush cultivars: ‘Arcadia’ and ‘Jewel’, respectively for the management of chilli thrips in 2019. The treatments and rates were AptaTM 15SC (tolfenpyrad, IRAC code #21A) at 27 oz/acre, Assail 30SG (acetamiprid, IRAC code #4A) at 5.3 oz/acre, Delegate WG (spinetoram, IRAC code #5) at 7 oz/acre, Sivanto Prime (flupyradifurone, IRAC code #4D) at 14 oz/acre, and Sivanto Prime with Induce (a nonionic adjuvant, 0.25% v:v).
Results are published in AMT. Link to pdf
Advisor: Sriyanka Lahiri, Assistant Professor (University of Florida)
Several insecticides were compared for efficacy in the field for management of chilli thrips at the research facilities of the Gulf Coast Research and Education Center, University of Florida, Wimauma, FL in 2019.
Products tested: Apta (tolfenpyrad, IRAC code #21A) at 27 oz/acre, Exirel (cyantraniliprole, IRAC code #28) at 16 oz/acre (low) and 20.5 oz/acre (high), Minecto Pro (abamectin, IRAC code #6 and cyantraniliprole, IRAC code #28) at 10 oz/acre, and Radiant (spinetoram, IRAC code #5) at 10 oz/acre. All experimental products were tank mixed with a surfactant, Induce, at the rate of 0.25% (v/v).
Results are published in AMT. Link to pdf
Blueberry (2019)
Advisor: Oscar Liburd, Professor (University of Florida)
Several insecticides were compared for the efficacy in two blueberry fields, located in Plant City, FL and Clermont, FL with southern highbush cultivars: ‘Arcadia’ and ‘Jewel’, respectively for the management of chilli thrips in 2019. The treatments and rates were AptaTM 15SC (tolfenpyrad, IRAC code #21A) at 27 oz/acre, Assail 30SG (acetamiprid, IRAC code #4A) at 5.3 oz/acre, Delegate WG (spinetoram, IRAC code #5) at 7 oz/acre, Sivanto Prime (flupyradifurone, IRAC code #4D) at 14 oz/acre, and Sivanto Prime with Induce (a nonionic adjuvant, 0.25% v:v).
Results are published in AMT. Link to pdf
Green peach aphid
|
Green peach aphid (Myzus persicae (Sulzer), Hemiptera: Aphididae) (2014)
Advisor: Dr. Dakshina Seal, Associate Scientist (University of Florida) Leaf dip bioassays were conducted to test the efficacy of several insecticides against green peach aphid in vegetable IPM laboratory, Tropical Research and Education Center, Homestead, FL. Products tested: Exirel, Actara, Provado, Movento, Obron, Beleaf, and Asana. |
cabbage aphid
|
Cabbage aphid (Brevicoryne brassicae L., Homiptera: Aphididae) (2012)
Supervisor: Mr. Rambabu Paneru, Scientist (Entomology Division, Nepal Agricultural Research Council) The experiment was carried out in field plots of cauliflower (Brassica oleracea L., Brassicaceae) at Entomology Division, Nepal Agricultural Research Council, Khumaltar, NP during 2012. Products tested with dose rate: JIWATU (cow urine and water @ 1:3) @ 50 ml/L water, Servo @ 15 ml/L water, Datura stramonium L. (Solanaceae) extract with water @ 1:1, Margosom (Niconim) @ 5 ml/L water, and Thiodan (Alden) @ 1.5 ml/L water. |
Grain weevil and moth
|
Grain weevil (Sitophilus oryzae L. and Sitophilus zeamais Motschulsky, Coleoptera: Curculionidae) and grain moth (Sitotroga cerealella Oliver, Lepidoptera: Gelechiidae) (2012)
Supervisor: Mr. Rambabu Paneru, Scientist (Entomology Division, Nepal Agricultural Research Council) The experiment was carried out at Entomology Division, Nepal Agricultural Research Council, Khumaltar, NP in locally adopted storage structure of maize i.e. BHAKARI under the local variety of maize (Zea mays L., Poaceae) in 2012. Different plant parts like leaves, twigs, seed, and rhizome of different botanicals were collected, shade dried, and grinded to powder. The grinded powder were then thoroughly mixed with each 17 Kg of maize for each BHAKARI at a rate of respective doses per kg of maize grains. Plants used: Jatropha curcas L., Euphorbiaceae (30 g), Solanum virginianum L., Solanaceae (30 g), Gaultheria fragrantissima, Ericaceae (50 g), Trigonella foenum-graecum L., Fabaceae (20 g), Solanum lycopersicum L., Solanaceae (30 g), Datura stramonium L., Solanaceae (30 g), Agave americana L., Agacaceae (30 g), Zingiber officinale Roscoe, Zingiberaceae (20 g), and Thymus vulgaris L., Lamiaceae (20 g). |
Epilachna beetle
|
Management of epilachna beetle, Epilachna vigintioctopunctata (Fab.) (Coleoptera: Coccinellidae) in bitter gourd, Momordica charantia L. (Cucurbitaceae) with synthetic and biological insecticides. (2011)
Supervisor: Mr. Binod Hamal, Senior Plant Protection Officer (Government of Nepal) Epilachna beetle is serious pest of cucurbitaceae in South Asia. Field plot study was conducted at research facility of Institute of Agriculture and Animal Science, Tribhuvan University, Rampur, NP during 2011 to test the efficacy of several commercial pesticide products against epilachna beetle in bitter gourd. Products tested: Derisom, Borer Guard, Metarhizium, Margosom, and Imidacloprid |